Electroplating uses an electrochemical reaction to place a controlled metal layer on a conductive part or on a surface that has been activated. A direct current drives metal ions in the solution to move and settle on the part, which improves the appearance, boosts corrosion resistance, increases surface hardness, strengthens conductivity, and adds other useful functions.
This guide walks you through how electroplating works, the different types you can choose from, the materials that fit this process, the surface results you can achieve, and the industries that rely on it. It also highlights the key differences between electroplating and electroforming so engineers, manufacturers, and hobbyists can fully understand this important finishing method.
How Does Electroplating Work?
Electroplating uses an electrolytic reaction to drive metal ions onto the part with direct current. The part works as the cathode, while the plating metal or an inert material works as the anode. Both sit in a solution that holds metal ions. When the power turns on, the anode releases metal ions. These ions move toward the cathode and gain electrons, then turn into solid metal on the surface, completing the core steps of the plating process.
Before plating starts, the part needs careful cleaning and activation because oil, oxide, or dirt will reduce adhesion. The uniformity, thickness, and quality of the coating depend on many factors such as current density, solution formula, bath temperature, additives, and the shape and position of the part. The coating grows as current and plating time increase. By adjusting these conditions, you can create layers that meet corrosion, appearance, or functional requirements throughout the plating process.
What are The Different Types of Electroplating Surface Treatments?
Electroplating includes many types based on the metal or alloy you choose. Each type brings specific benefits such as corrosion protection, electrical performance, hardness, gloss, and cost control.
Below are the most common electroplating methods:
Nickel Plating
Nickel plating improves corrosion resistance, hardness, and wear strength while also giving a clean finish. The nickel layer looks gray and dense. It works as a functional coating and often serves as a base for other metals to improve adhesion and solderability.
You can choose bright nickel for a shiny look or electroless nickel for even thickness on complex shapes. Nickel plating is common in mechanical parts, electronic connectors, automotive parts, and decorative components.
Zinc Plating
Zinc plating gives steel parts cost-effective corrosion protection. The zinc layer offers sacrificial protection, which means zinc reacts first and slows rust on the steel even in humid or harsh conditions.
Many industries use zinc plating for fasteners, hardware, and automotive parts. You can add clear, yellow, or black chromate treatment to improve corrosion resistance and change the appearance.
Copper Plating
Copper plating gives excellent conductivity and smooth surface quality, so many electronic and precision parts use it. Copper fills small surface flaws and creates a strong foundation for nickel, gold, or silver. It often works as the base layer in multi-step plating.
Copper plating appears in electrical connectors, printed circuit boards, and parts that need stable electrical flow.
Chromium Plating
Chromium plating creates a hard, bright, and wear-resistant surface. It serves both decorative and functional needs.
Decorative chrome is thin and usually sits on top of a nickel layer. It gives faucets, trims, and many consumer items a mirror-like finish. Hard chrome is thicker and works in industrial parts such as piston rods and molds to improve wear strength and corrosion performance.
Chrome resists scratches, oxidation, and color change, so it stays popular in automotive, tool, and high-durability products.
Gold Plating
Gold plating shines in applications that demand top electrical performance and long-term stability. The gold layer prevents oxidation, keeps low contact resistance, and holds steady over time. Many industries rely on it for electronic contacts, aerospace parts, and high-end devices.
Since gold is expensive, most applications use a thin layer that still provides strong performance. Gold plating also gives a bright and premium appearance, so it remains a favorite in jewelry and luxury goods.
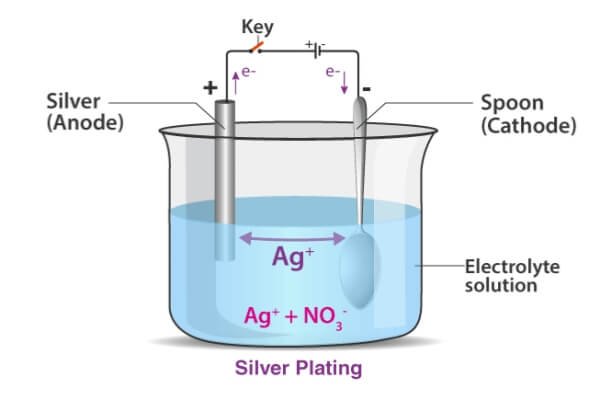
Silver Plating
Silver plating offers the highest electrical and thermal conductivity among common metals. It also has a bright, reflective surface.
Manufacturers use silver plating for RF parts, electrical contacts, connectors, and reflective components. It improves electrical performance but can lose its shine in the air, so many products add a protective coating or extra finishing. Its electrical and optical qualities make it ideal for high-frequency electronics, precision components, and premium decorative items.
Tin Plating
Tin plating gives a safe, solder-friendly, and corrosion-resistant surface, so it plays a big role in electronics and food-related products. Tin stops oxidation on the base metal and supports easy soldering for connectors, terminals, and circuit boards.
Because tin stays stable when it touches food, many food containers use tin plating to protect contents and extend shelf life. Its mix of safety and performance makes tin plating important in electronics and packaging.
Palladium Plating
Palladium plating gives high hardness, wear resistance, and strong corrosion protection. Many view it as a durable alternative to gold.
Palladium provides steady electrical performance and long-term reliability, so it works well in connectors, sensors, medical parts, and other high-performance components. It also resists discoloration and offers mild catalytic behavior, which makes it useful in jewelry and premium parts.
Alloy Plating
Alloy plating combines two or more metals to create a coating with blended properties. Tin-nickel and zinc-nickel improve corrosion resistance and strength. Copper-zinc works well in decorative pieces. Nickel-iron suits magnetic applications.
By choosing different metal combinations, manufacturers can tailor hardness, durability, or color to meet both functional and visual needs.
What Plastic Materials Are Compatible with Electroplating?
Some plastics gain strong adhesion and good conductivity after surface activation or chemical treatment. Their suitability depends on how well the plastic bonds with metal and whether it can handle the conditions during electroplating. Common options include ABS, polycarbonate, polypropylene, polyethylene, nylon, and PEEK.
With proper preparation, these plastics form a smooth and durable plated surface that works well in electronics, automotive parts, decorative items, and industrial components.

Acrylonitrile Butadiene Styrene (ABS)
ABS is one of the most common electroplating plastic materials because its structure allows easy surface treatment and strong bonding. The butadiene in ABS creates a rough micro surface after acid etching, which gives the metal layer a firm base. Many industries use plated ABS for car interior parts, toys, and electronic housings that need a decorative or functional metal look.
Polycarbonate (PC)
Polycarbonate offers good heat resistance and strong impact performance. After the right surface treatment, it holds metal coatings well. PC often needs chemical etching or plasma activation to improve adhesion.
Plated PC appears in car lighting parts, electronic housings, and medical devices. It gives products a clean metal shine and stable mechanical strength.
Polypropylene (PP) and Polyethylene (PE)
PP and PE have nonpolar surfaces, so they do not bond well with metal at first. They need strong surface activation before electroplating.
Common steps include flame treatment or plasma treatment to create active points for better adhesion. Treated PP and PE work well in chemical containers, outdoor tools, and other products that need low weight, chemical resistance, and metal protection.
Nylon (Polyamide)
Nylon provides high strength and good heat resistance, so many industrial parts use it.
It needs chemical etching or other surface conditioning to improve bonding with metal during electroplating.
Plated nylon appears in gears, valves, fasteners, and aerospace parts. It adds corrosion protection and strength while keeping the material light.
Polyetheretherketone (PEEK)
PEEK is a high-performance plastic that handles extreme heat and chemical exposure. It fits well for metal coating after proper treatment.
It often needs plasma or laser preparation to improve adhesion.
Plated PEEK supports aerospace parts, medical tools, oil and gas equipment, and high-temperature electronics. It offers long-term stability, heat resistance, and strong corrosion protection.
What Are the Key Benefits of Electroplating?
Electroplating brings both functional and visual advantages, which make it valuable in manufacturing and decorative work.
1. Stronger corrosion resistance
Many coatings, such as zinc or nickel, create a solid barrier that stops the base metal from reacting with moisture or chemicals. Parts last longer in outdoor or harsh environments.
2. Better appearance
Chrome, gold, or silver creates clean, bright, and consistent surfaces. These finishes make consumer goods, jewelry, and decorative parts more attractive.
3. Improved performance
Each metal adds a unique property. Gold and silver support strong conductivity, chrome increases hardness, and tin improves solderability. By choosing the right metal, manufacturers can match performance to the final use.
4. Higher cost efficiency
A thin layer of a precious metal can deliver excellent results while keeping the base material inexpensive. This approach saves cost without sacrificing quality.
5. Extended part life through repair
Thin metal layers can fill worn areas or restore damaged surfaces. Many industries use electroplating to repair tools, shafts, and precision parts.
6. Precise control of coating thickness
Engineers can adjust current and time to achieve exact thickness levels. This flexibility supports both decorative finishes and strict functional requirements.
What Are the Limitations of Electroplating?
Electroplating offers many advantages, but it also comes with several limitations that you need to consider.
1. Environmental and safety risks
Many plating solutions contain toxic chemicals such as cyanides or heavy metals. Facilities must manage waste liquid and wastewater carefully to meet environmental rules.
2. Thickness and geometry challenges
Most coatings stay within the micrometer range. Very thick layers may crack or peel. Complex shapes, such as deep holes or sharp corners, also make uniform plating difficult. High-quality results often require advanced tools and strong process control.
3. Material restrictions and higher operating costs
Electroplating only works on conductive materials or on surfaces that have been activated. Maintaining solution chemistry, cleaning parts, and running the power supply add cost, especially in small production runs.
4. Compatibility issues
Some metal combinations may cause problems such as galvanic corrosion or hydrogen embrittlement, which can reduce long-term reliability.
What Are The Common Methods of Metal Electroplating?
Metal plating can be done through several methods, each with unique strengths for different sizes, shapes, and quality needs. Common methods include rack plating, barrel plating, brush plating, and pulse plating.
The best choice depends on part size, quantity, shape, and the required performance.
Rack Plating
Rack plating places the parts on metal fixtures or hooks to keep them stable in the solution. This method controls angles and positions well, which helps create a consistent coating. It works best for large, delicate, or high-value parts.
Rack plating supports small and medium batch sizes and offers strong quality in both appearance and performance.

Barrel Plating
Barrel plating handles large batches of small parts such as screws, nuts, and washers. The rotating barrel keeps the parts moving so each one contacts the solution and receives a fairly even coating.
This process offers high efficiency and low cost. Since the parts rub against one another, it is not the best choice for items with delicate surfaces or high decorative requirements. It suits functional small parts.
Brush Plating
Brush plating uses a hand-held anode wrapped with an absorbent material soaked in solution. The operator moves the tool back and forth on the surface while current flows. There is no need to immerse the entire part.
This method works well for large equipment, fixed structures, or small repair areas. Technicians can restore a coating on-site without removing the part, which makes this a fast and flexible repair solution.

Pulse Plating
Pulse plating uses short bursts of current instead of a steady flow. These on-off cycles control how metal ions settle on the part. The result is a coating that is more uniform, dense, and smooth.
This method improves hardness, adhesion, and corrosion resistance. It also handles complex shapes well. Many industries use pulse plating for electronic parts and aerospace components. It does require special power supplies to keep the pulse pattern stable.
What Color Options Are Available in Electroplating?
Electroplating offers a wide range of stable color finishes by selecting different metals, alloy formulas, and post-treatment methods.
Common choices include bright silver tones from nickel, chromium, or rhodium plating, which create a clean metallic shine.
Gold finishes vary from deep yellow to soft rose gold depending on the purity and alloy blend.
Black shades come from black nickel or black chrome, offering either a soft matte look or a deep glossy finish.
Warm colors such as copper or bronze work well for decorative parts. With specific additives or dyes, the plating bath can also create custom colors like blue, green, or even purple.
Where Is Electroplating Used?
Electroplating serves many industries because it improves corrosion resistance, conductivity, and appearance.
Aerospace: It boosts the reliability of electrical contacts and protects vital components from harsh environments.
Automotive: Zinc and chrome plating prevent rust on fasteners and enhance the look of trims.
Jewelry: Gold, silver, and rhodium plating create stable colors and polished finishes while reducing material cost.
Art and Home Décor: Faucets, sculptures, and lighting parts use electroplating to achieve modern or classic metal textures.
Dental and Medical: Plating strengthens tools, crowns, and brackets while improving corrosion resistance and biocompatibility.
Electroforming vs Electroplating
Electroforming and electroplating both rely on electrodeposited metal, but their goals and methods differ.
Electroplating adds a thin metallic layer—usually only a few micrometers—to a part to improve corrosion resistance, conductivity, or visual appeal. It does not change the part’s shape.
Electroforming builds a much thicker metal layer on a temporary mold, which is then removed to create a standalone metal part. This method works well for micro-components, jewelry molds, and precision waveguides. For an in-depth look at how electroforming differs and its industrial applications, see ScienceDirect: Electroforming Overview.
The processes also differ in operation. Electroplating uses higher current density and shorter processing time with a strong focus on adhesion. Electroforming runs with lower current and longer deposition time to create a self-supporting structure.
In short, electroplating enhances an existing part’s surface, while electroforming creates an entirely new metal component.
Conclusion
Electroplating is a flexible surface-finishing method that balances performance and appearance. Through electrochemical deposition, metal or plastic parts gain stronger corrosion resistance, higher hardness, better conductivity, and improved visual appeal. Many manufacturers rely on professional electroplating services to achieve consistent quality across different materials and product designs.
Whether you choose nickel, gold, chrome, or alloy plating, each option provides unique performance and color characteristics. Hanging plating, barrel plating, brush plating, and pulse plating further expand the range of possible applications.
Despite limitations such as environmental control, thickness uniformity, and material compatibility, electroplating remains essential in aerospace, automotive, jewelry, medical devices, and home décor. Compared with electroforming, electroplating focuses on upgrading a part’s surface, making it a key process for product protection, long-term durability, and premium appearance.
For professional electroplating services or custom-plated metal parts, contact HRCCNC today. Our team can help you choose the right materials, colors, and plating methods for your project. Get a quote now and see how we can enhance your parts’ performance and appearance.





