You have to notice the surprising weight whenever you pick up a lead (Pb) chunk. The ordinary metal feels far heavier than many other common elements.
But what makes lead so heavy? The answer lies in lead (Pb) density. Let’s explore the values, factors, and applications to understand why lead is so heavy.
What is the Density of Lead (Pb)?
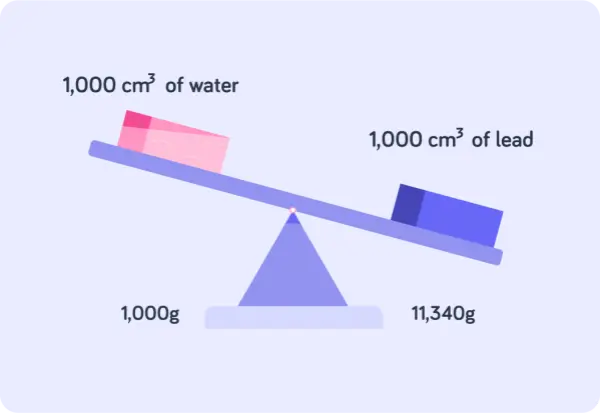
Pure lead’s density is around 11.34 g/cm³ or 11,340 kg/m³. It’s one of the heaviest elements found in industry and consumer applications.
From a scientific perspective, density (ρ) is the mass (m) per unit volume (V).
ρ = m / V
Therefore, a cube of 1cm3 made of pure lead weighs 11.34 grams. It means even a small solid (volume) of lead weighs a great amount (mass).
Lead atoms have a heavy nucleus that contributes to their density. You may wonder how heavy lead is compared to other common metals. –
- Aluminum (Al) = 2.70 g/cm³
- Iron (Fe) = 7.87 g/cm³
- Standard Steel = 7.85 g/cm³
- Copper (Cu) = 8.96 g/cm³
- Gold (Au) = 19.32 g/cm³
- Silver (Ag) = 10.49 g/cm³
- Osmium (Os) = 22.59 g/cm³
- Bismuth (Bi) = 9.78 g/cm³
- Tungsten (W) = 19.25 g/cm³
Example: A lead brick (10 cm × 10 cm × 10 cm) weighs about 11.34 kg. Meanwhile, the same-sized aluminum brick weighs only 2.7 kg.
Lead Density Conversion Table (Different Units)
The international systems (SI) defines g/cm³ as the unit to express material density. However, scientific or industrial applications may require conversion to different units.
| Measurement Unit | Lead Density | Context/Application |
| g/cm³ (gram per cubic centimeter) | 11.34 | Chemistry, materials science |
| kg/m³ (kilogram per cubic meter) | 11,340 | Engineering, construction |
| lb/ft³ (pound per cubic foot) | 708 | Civil engineering, US customary units |
| lb/in³ (pound per cubic inch) | 0.41 | Manufacturing, machining |
| g/mL (gram per milliliter) | 11.34 | Laboratory experiments |
| ton/m³ (ton per cubic meter) | 11.34 | Mining, shipping |
| oz/in³ (ounce per cubic inch) | 6.57 | Jewelry, small-scale crafts |
Why is Lead So Dense?
Simply put, lead feels heavy because its atoms carry a lot of mass. The atoms also pack together rather efficiently in a solid.
In total, high atomic mass per atom + close atomic packing = high mass per unit volume. That’s why the density at room temperature is about 11.34 g/cm³
Factors Affecting the Density of Lead
Lead’s cited density at room temperature may slightly alter. The value can vary depending on physical conditions, chemical composition, and structural factors.
Understanding the variations is crucial for CNC machining and custom metal component manufacturing. It’s because density directly impacts tolerances, machining speeds, and final product performance.
● Atomic Number
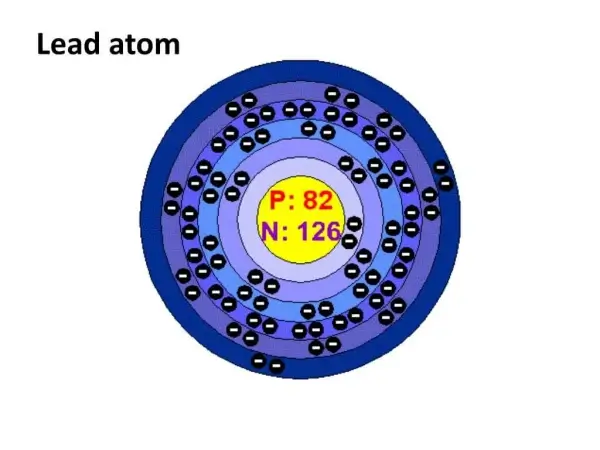
Lead’s nucleus contains 82 protons and a large number of neutrons. The pileup makes each atom intrinsically massive compared to lightweight metals like aluminum or iron.
● Atomic Mass
One lead atom has a standard atomic weight of 207.2 Atomic Mass Unit (AMU). So, even a modest volume can contain many grams of mass. And custom component manufacturing requires special handling and fixturing during CNC operations.
● Crystal Structure
Lead crystallizes in a face-centered cubic (FCC) lattice. And FCC is one of the densest ways to arrange spheres. The packing arrangement can fill up to 74% of available spaces with atoms.
● Thermal Expansion
Metal density slightly decreases due to thermal expansion resulting from increased temperatures. Lead has a thermal expansion coefficient of 29 × 10⁻⁶ /K (inverse Kelvin).
Example: Lead density (standard) is 11.34 g/cm³ at 20°C, whereas it’s 11.28 g/cm³ (slightly lower) at 100°C. Temperature shifts can alter tolerances in precision machining.
CNC shops maintain climate-controlled environments to guarantee accuracy in lead-based or alloyed components.
Lead-based coolant systems in nuclear reactors must keep the density variations with temperature in mind for safe heat transfer.
● Pressure
High pressure forces the atoms closer, increasing density. Several gigapascals (GPa) can increase lead’s density by 1% – 2%. However, they’re more relevant for material science experiments. Pressure-based processes like forging or powder compaction can slightly alter density, which machinists must consider when cutting or finishing parts.
● Purity and Alloying
The numeric value for lead density belongs to pure lead only. Alloys (lead-tin solder or lead-antimony batteries) have slightly different densities.
Example: Lead-antimony alloy in car batteries has a density of 10.7 to 11.2 g/cm³. Meanwhile, lead-tin solder features a lower density range of 8.5 to 9.0 g/cm³.
Appropriate alloying reduces density while improving the mechanical strength. That’s why lead alloys are more practical for manufacturing purposes.
● Isotopic Composition
Natural lead consists of four stable isotopes: Pb-204, Pb-206, Pb-207, Pb-208. And Pb-208 is the most abundant (52.4%).
Slight variations in isotopic ratios can cause minuscule (negligible for everyday use) differences. It’s more suitable for geophysical studies.
● Microstructural Factors
Porosity: Lead products with microscopic voids (castings with air bubbles) have lower effective density.
Grain size: Fine-grained lead tends to pack more efficiently, slightly increasing density.
Example: Controlled porosity in die casting enables consistent density for radiation shielding panels.
Porosity and grain size must be considered in CNC machining and precision manufacturing to control the cutting behavior, tool wear, and surface finish. Shops specializing in custom lead components perform microstructural analysis before machining to guarantee consistency.
Real-World Applications: High-Density Lead
● Medical Imaging
X-ray and CT scan rooms worldwide use lead sheets to block radiation. A 2 mm thick lead sheet weighs 22.7 kg/m². It provides effective shielding without excessive thickness.
Those sheets must be precision-machined and cut to exact dimensions to fit seamlessly into equipment. A perfect fit also ensures safety and compliance with medical standards.
● Nuclear Reactors
Lead is used as a coolant and shielding material in advanced reactors. Its density and atomic number (82) make it highly effective at absorbing gamma rays.
Reactor shielding blocks and coolant system components call for custom machining and tight tolerances. And CNC processes guarantee reliability under extreme conditions.
● Car Batteries
Lead batteries are still the most widely used rechargeable battery type. Each battery contains 10 – 15 kg of lead. Such mass contributes to massive weight while ensuring reliability.
Lead grids and plates inside batteries are manufactured through precision machining + alloy processing. It becomes easy to hold consistent density and electrochemical performance across millions of units.
● Ships and Submarines
Lead ballast provides stability by concentrating weight in small volumes. A submarine may use several tons of lead ballast to maintain buoyancy control.
Ballast blocks must be custom-fabricated and machined to exact specifications. Otherwise, they can’t integrate smoothly into vessel designs without compromising structural integrity.
● Construction/Infrastructure
Metal sheets (lead) are installed across strategic locations in buildings to reduce noise transmission. Its density helps absorb sound waves and vibrations.
Machined lead panels in construction are cut to precise sizes for architectural integration. You can enjoy effective soundproofing and vibration damping.
● Ammunition in Defense
Lead’s density ensures compact, high-mass projectiles. Alloying with antimony induces hardness; lead remains the core of most ammunition.
Ammunition manufacturing relies on precision machining and forming processes. It enables uniform projectiles with consistent ballistic performance.
● Art, Sculpture, and Historic Uses
Lead strips (“cames”) hold colored glass pieces together in cathedrals worldwide. The high density provides structural stability while remaining malleable.
Even in artistic applications, custom machining and shaping of lead strips ensure durability. And then there’s an exact fit for intricate stained-glass designs.
● Scientific + Industrial Uses
Lead bricks line experimental labs handling radioactive materials. Precision instruments, elevators, and cranes to accommodate the compact counterweights are requirements.
Those bricks and counterweights come from CNC machining and precision fabrication. Thus, they ensure uniform density and dimensional accuracy for critical industrial applications.

How to Measure the Density of Lead?
a. Direct Measurement Method (Mass ÷ Volume)
Step 1: Measure Mass
Use a digital balance to weigh a lead sample. For example, you have a lead cube weighing 92.1 g.
Step 2: Measure Volume
If the sample is a regular shape (cube or cylinder), calculate volume using geometric formulas. For a cube of 2 cm sides, V = 23 = 8 cm3.
Step 3: Calculate Density
Density = 92.1 / 8 = 11.51 g/cm3. It’s slightly higher than the standard due to measurement error, showing why precision matters.
b. Water Displacement Method (Archimedes’ Principle)
- Step 1: Fill a graduated cylinder with water and record the initial volume.
- Step 2: Submerge the lead object and record the new water level.
- Step 3: The difference between the levels represents the volume of the sample.
- Step 4: Divide the lead sample’s mass by the displaced volume.
Example: You have a lead fishing sinker weighing 50g. The water level rises by 4.41 mL when submerged.
Density = (50 ÷ 4.41) ≈ 11.34 g/cm3. However, it matches the standard density perfectly. That’s why it’s the go-to method for mining and metallurgy labs.
c. Pycnometer Method (High Precision Laboratory Technique)
A pycnometer is a specialized flask used to measure the density of liquids and solids. It’s suitable for lead powder (finely divided samples).
Fill the pycnometer with a liquid of known density to calculate the displacement. Its accuracy is incredibly high, as much as ±0.01 g/cm³.
d. Industrial and Engineering Methods
- X-ray or Ultrasonic Techniques: Checking density uniformity in lead shielding panels.
- Hydrostatic Weighing: Weighing in air and then in water to calculate the buoyant force.

Toxic Hazards of Lead: Safe Usage with Environmental Compliance
Lead isn’t just heavy; it’s also hazardous. Therefore, addressing its toxicity for safe handling and compliance is mandatory to ensure its credibility in sectors like medical, nuclear, and automotive.
Toxicity of Lead
Children and pregnant women are especially vulnerable to lead’s neurotoxic effects. Harm can occur even at relatively low exposure. Lead is a cumulative toxicant that affects the nervous, hematological, gastrointestinal, cardiovascular, and renal systems.
- Exposure Pathways: Inhalation of dust/fumes during melting, machining, or recycling; ingestion via hand-to-mouth contact; exposure from legacy paints, plumbing, and soils.
- High-Risk Operations: Battery manufacturing and recycling, smelting, casting, radiation-shield installation and cutting; demolition of legacy lead-containing infrastructure.
- Enterprise Risk: Occupational exposure has long been recognized (historically documented “lead colic”); modern supply chains still rely heavily on secondary refining of battery scrap.
Safe Usage Requirements
Implementation of a layered control is obligatory. Engineering controls followed by administrative controls, then PPE, and regular inspection should work in the field.
a. Engineering Controls
Local exhaust ventilation captures fumes/dust during melting, soldering, and cutting. Use closed transfer systems for lead powders and pellets; ventilated booths for high-emission tasks. Replace dry sweeping with wet cleaning; get certified HEPA vacuuming to avoid redistributing dust.
b. Administrative Controls
Limit time in high-fume areas; rotate tasks to reduce cumulative dose. Prohibit food/drink in work areas; mandate handwashing before breaks; allocate dedicated changing rooms + on-site showers. Conduct wipe tests in critical zones; sealed containers for waste; controlled laundry services for workwear.
c. Personal Protective Equipment (PPE)
Invest in professionally fitted respirators for tasks with potential airborne lead. Align the cartridge selection based on the contaminants generated. Get disposable coveralls, gloves rated for chemical and abrasion resistance, and shoe covers against cross-contamination. Don’t forget eye/face protection.
Environmental Compliance Essentials
Regulators expect proactive compliance instead of show-off paperwork. Robust systems must prevent releases, reduce risk to communities, and document performance transparently.
- EPA (United States): Emphasizes compliance assistance, auditing, and self-disclosure programs. Encouraging organizations to assess obligations and fix issues swiftly.
- Occupational Safety Oversight: Sector-specific guidance for smelting, mining, and battery recycling. Supports hazard mitigation and enforcement, reflecting the industry’s scale and risk profile.
- Public Health Guidance: WHO underscores the need for population-level prevention. Recognizes the severe societal costs of lead exposure and advocates policies against contamination.
Conclusion
In summary, the high density of lead is a unique physical property that also renders it irreplaceable in numerous critical fields. It can be for radiation shielding in medical equipment, or for storage batteries requiring high energy density. No matter the case, precise control over material density directly determines the performance and reliability of the final product.
Standard and Custom Parts Machining with Precision at HRCCNC
At HRCCNC, we possess in-depth expertise in the correlation between material properties and processing technologies. We offer a full range of lead and lead alloy materials that comply with ASTM standards. Our 18 years of precision machining experience help you translate the advantages of high-density lead high-performance end components.
Need to select the right lead alloy for your project or customize high-precision lead components? Our team of material and processing experts is ready to provide support at any time.





