‘Does aluminum rust like steel/iron?’ – the simple question pops up more often. Anyone working with aluminum siding, cookware, or industrial components has to worry about rust.
However, the metal doesn’t rust in the traditional sense. Instead, aluminum undergoes oxidation that leads to corrosion. This article breaks down the nature of corrosion in aluminum.
Rust vs Corrosion: What’s the Difference?
The terms rust and corrosion have been used interchangeably in metal degradation. They’re not the same. The distinction is fundamental when evaluating materials like aluminum.
What Is Rust?
It’s a specific corrosion type that occurs only with iron and iron-based alloys like steel. Rust results from a chemical reaction between iron, oxygen, and moisture. It’s the formation of iron oxide (Fe₂O₃) on the surface. The chemical reaction –
4Fe + 3O2 + 6H2O → 4Fe(OH)3 → Fe2O + 3·xH2O
The hydrated iron oxide [Fe2O + 3·xH2O] eventually weakens the metal structure. Rust mostly appears as reddish-brown flakes/patches that expand and crumble while exposing more metal to degradation.
Bridges, pipelines, and vehicles made of untreated steel show visible rust damage. The collapse of the Silver Bridge (1967) in West Virginia was partly attributed to rust-induced structural failure.

What Is Corrosion?
It’s a broader or umbrella term that refers to the gradual degradation of material. Corrosion may occur through chemical or electrochemical reactions with the environment. It includes metals like Al, Cu, Zn, and even non-metals like polymers and ceramics.
Notably, reactive aluminum (Al) forms a thin layer of aluminum oxide (Al₂O₃) on the surface. Unlike rust, the layer is considerably stable and protective from the start. The oxide layer prevents further oxidation unless disrupted. The chemical reaction –
4Al + 3O2 → 2Al2O3
Surprisingly, there are distinctive types of corrosion that affect aluminum. The aluminum product may be subjected to pitting, galvanic, and/or filiform corrosion.
Aircraft fuselages rely on aluminum alloys that resist corrosion while remaining lightweight. However, they still require protective coatings + regular inspections to prevent pitting and galvanic corrosion.
Can Aluminum Rust?
Aluminum does not rust.Rust, by definition, is iron oxide (Fe₂O₃). The flaky compound forms when iron/steel reacts with oxygen and moisture. Since aluminum is a non-ferrous metal, it can’t produce rust under any circumstances.
The distinction goes well beyond semantics as it has real-world implications for everything. Steel bridges may require constant rust-proofing, whereas aluminum structures hold up with minimal intervention.
Aluminum’s immunity to rust isn’t a marketing gimmick. It comes from a metal’s iron-free composition. The nature is more or less the same across aluminum products, whether made from pure Al or Al alloys.
Does Aluminum Corrode?
Aluminum corrodes.Aluminum is well-received for its corrosion resistance. However, the material isn’t exactly immune. Instead, aluminum undergoes a different kind of degradation.
It forms a thin and protective layer of aluminum oxide (Al₂O₃) that shields the underlying metal. Even the layer gets compromised under varying conditions, leading to different forms of corrosion.

01. Pitting Corrosion
Chloride ions (from saltwater or industrial pollutants) penetrate the oxide layer. You’ll see small pits form on the surface. They’re often invisible until the pits deepen and compromise structural integrity.
Pitting corrosion mostly occurs across marine settings, coastal buildings, and industrial zones. Aluminum boat hulls and offshore platforms often suffer pitting unless treated with marine-grade coatings.
02. Galvanic Corrosion
Aluminum contacts a more noble metal (like Cu or stainless steel) in the presence of an electrolyte (water). Al acts as the anode and corrodes faster than it would alone.
Galvanic corrosion occurs across mixed-metal assemblies in humid or wet conditions. Improper joining of aluminum and titanium without insulation can lead to galvanic corrosion.
03. Crevice Corrosion
Moisture or chemicals are trapped in tight spaces like under gaskets, washers, or lap joints. It creates micro-environments where oxygen is depleted, accelerating localized corrosion.
Poorly ventilated or sealed structures, especially in humid climates, trigger the issue. Aluminum window frames in tropical regions often corrode at joints if not properly sealed.
However, there are over 13 recognized forms of corrosion in aluminum. Aside from pitting, galvanic, and crevice, you may have to deal with rarely occurring intergranular and stress corrosion cracking.
Factors to Trigger Corrosion in Aluminum
01. Air and Humidity
Aluminum performs exceptionally well in most atmospheric conditions. It reacts with oxygen upon air exposure to form a stable layer of aluminum oxide (Al₂O₃). The layer is –
- Self-healing: If scratched, it reforms almost instantly.
- Adherent: Unlike rust, it doesn’t flake off.
- Protective: It prevents further oxidation and corrosion.
Humidity levels influence the rate of oxide formation. However, aluminum remains unaffected mainly in moderate climates. Anodized aluminum in architecture/electronics relies on this principle for durability.
02. Freshwater Exposure
Aluminum fares well in freshwater environments, such as rivers, lakes, and rainwater. The oxide layer remains intact, and corrosion is typically slow and localized.
Low conductivity of freshwater reduces galvanic corrosion risk. Minimal chloride content means pitting is rare. However, stagnant water or water with high organic content can promote crevice corrosion.
03. Saltwater/Marine Environments
Saltwater is aluminum’s most aggressive adversary. The chloride ions in seawater penetrate the oxide layer, leading to pitting corrosion. You’ll see small but deep holes that compromise structural integrity.
Pitting is insidious and often goes unnoticed until failure occurs. Galvanic corrosion risk also increases when aluminum contacts other metals in wet conditions.
In coastal cities like Miami, USA, aluminum structures require marine-grade alloys (5083, 6061-T6). Even so, they need protective coatings to withstand salt-laden air and spray.
04. Industrial Environments
In industrial settings, aluminum faces threats from acidic fumes, alkalis, and chemical pollutants. These substances can –
- Disrupt the oxide layer, exposing raw aluminum.
- Accelerate corrosion, especially in poorly ventilated areas.
- Cause localized damage, such as crevice or intergranular corrosion.
In German and Chinese chemical plants, aluminum piping and panels are exposed to sulfur dioxide or hydrochloric acid. They require specialized coatings or alloy selection to prevent rapid degradation.

How to Protect Aluminum from Corrosion
Aluminum’s natural resistance to corrosion makes it a favorite across industries. But in aggressive places like coastal zones, industrial plants, or humid climates, even aluminum needs protection.
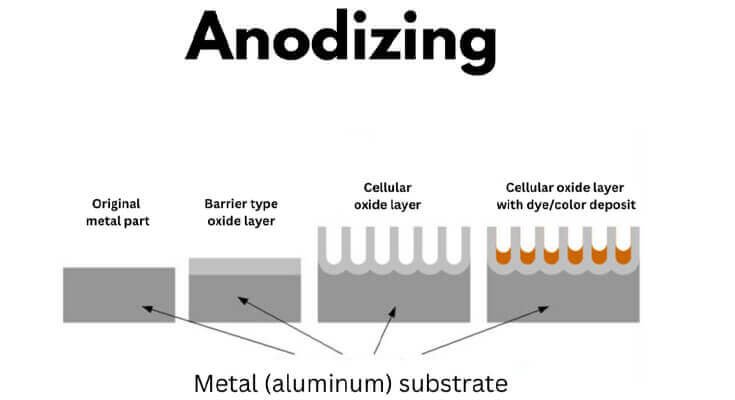
01. Anodizing
It’s an electrochemical process that thickens aluminum’s natural oxide layer. The surface becomes more durable, non-conductive, and resistant to corrosion.
Anodizing can increase the oxide thickness from 5 nm to 10 – 25 µm. It also enhances resistance to pitting and wear. You can induce vibrant yet fade-resistant coloring.
Anodized Al is widely used for signage, window frames, and consumer electronics in humid climates. Apple’s MacBook line uses anodized aluminum for both aesthetics and durability.
02. Painting or Powder Coating
These coatings act as physical barriers, preventing moisture, salts, and pollutants from reaching the aluminum surface.
- Painting: It’s ideal for large structures like bridges or buildings. The process requires surface preparation and periodic reapplication.
- Powder coating: It’s about the electrostatic application of dry powder, cured under heat. You’ll get a more durable and environmentally friendly outcome.
In coastal cities like Miami, powder-coated aluminum is used for railings, cladding, and marine equipment. They can effectively resist salt spray and UV degradation.
03. Alloying
Aluminum alloys combine aluminum with other elements (magnesium, silicon, zinc) to improve strength, corrosion resistance, and workability. Corrosion-resistant aluminum alloys –
- 5052: Excellent resistance to saltwater and marine environments.
- 6061-T6: Versatile, strong, and corrosion-resistant – widely used in structural applications.
- 5083: Marine-grade alloy with superior resistance to pitting and galvanic corrosion.
In shipbuilding across Southeast Asia, 5083 alloy is preferred for hulls and decks. The resistance to seawater corrosion and mechanical stress lends a hand.
04. Regular Maintenance
Use plastic or rubber insulators between dissimilar metals. Consider designing a drainage system to avoid trapped moisture. Apply sealants around joints and fasteners.
- Cleaning: Removes dirt, salts, and pollutants that can degrade the oxide layer or initiate crevice corrosion.
- Inspection: Identifies early signs of pitting or galvanic corrosion.
- Avoiding galvanic pairing: Prevents contact between aluminum and more noble metals (copper, stainless steel) in wet environments.
In industrial zones like Shenzhen, China, aluminum components in electronics and machinery are routinely cleaned and insulated. It prevents galvanic corrosion from adjacent copper wiring.
Conclusion
Aluminum never rusts like iron or steel. However, such a fact doesn’t make the material invincible. Salty, humid, or chemically aggressive environments can initiate corrosion. A rightful preventive measure can turn aluminum into one of the most durable choices.
Get the Best Corrosion-Free Aluminum from HRC
Don’t let the adverse conditions degrade your product. HRC specializes in protective aluminum making for 17+ years with practical and advanced solutions. Contact us for more on our CNC machining aluminum parts service.





